Heart Function and Related Conditions
Heart failure rarely occurs on its own. Various conditions can make the heart work harder than it should, exacerbating your heart failure symptoms. Heart failure can also lead to additional complications, such as kidney or liver disease or heart valve difficulties.
As part of confirming your diagnosis, your doctor will likely order blood tests and other testing to examine what’s wrong with your heart and to rule out any other diseases.
Coronary Artery Disease
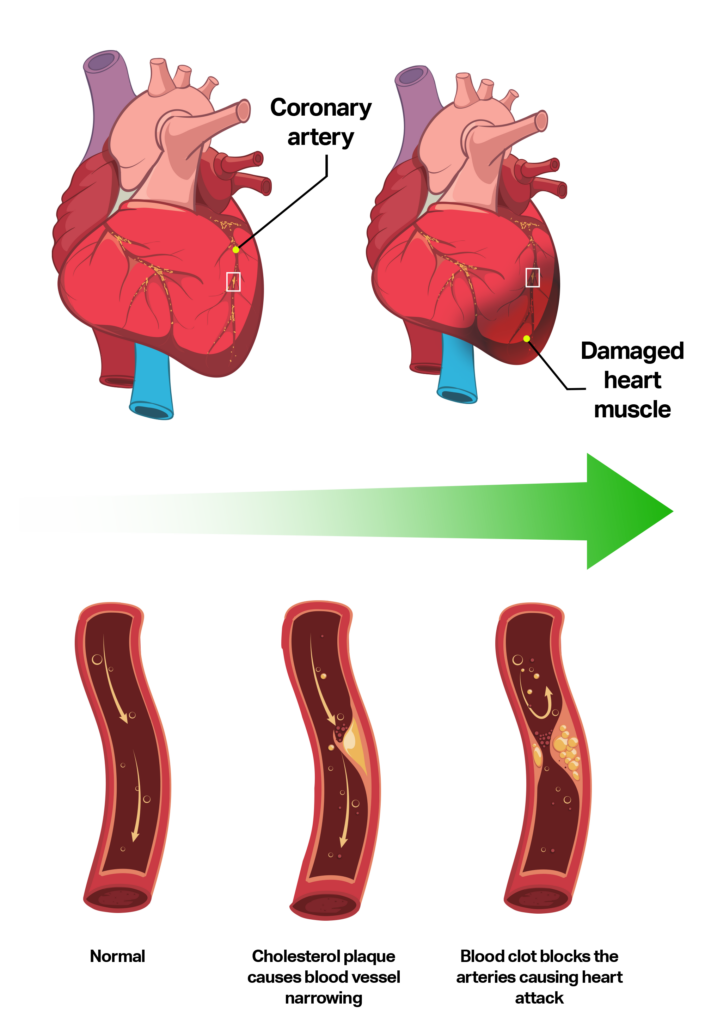
It occurs when cholesterol deposits in the walls of the coronary arteries, causing them to become narrowed or blocked. This deposit of cholesterol plaques is called atherosclerosis.
Over time, coronary arteries may become too narrow and impede blood flow to the heart muscle. This may result in chest pain (called angina), cause a heart attack, and weakened heart muscle.
A heart attack, or ‘myocardial infarction occurs when the cholesterol plaques spontaneously rupture, causing a blood clot to form within the artery. This results in a sudden lack of blood flow and oxygen to the heart muscle. It is not entirely clear what causes certain cholesterol plaques to rupture.
Coronary disease is typically diagnosed through different types of stress tests. The stress may either be exercise on a treadmill or bicycle, or a ‘pharmacologic stress’ brought on by medication.
To learn more about cardiovascular diagnostic tests
Risk factors for cholesterol plaques include:
- High blood pressure
- High cholesterol
- Diabetes
- Smoking
- Older age
- Inactivity
- Obesity
- Family history of heart disease.
Atrial Fibrillation
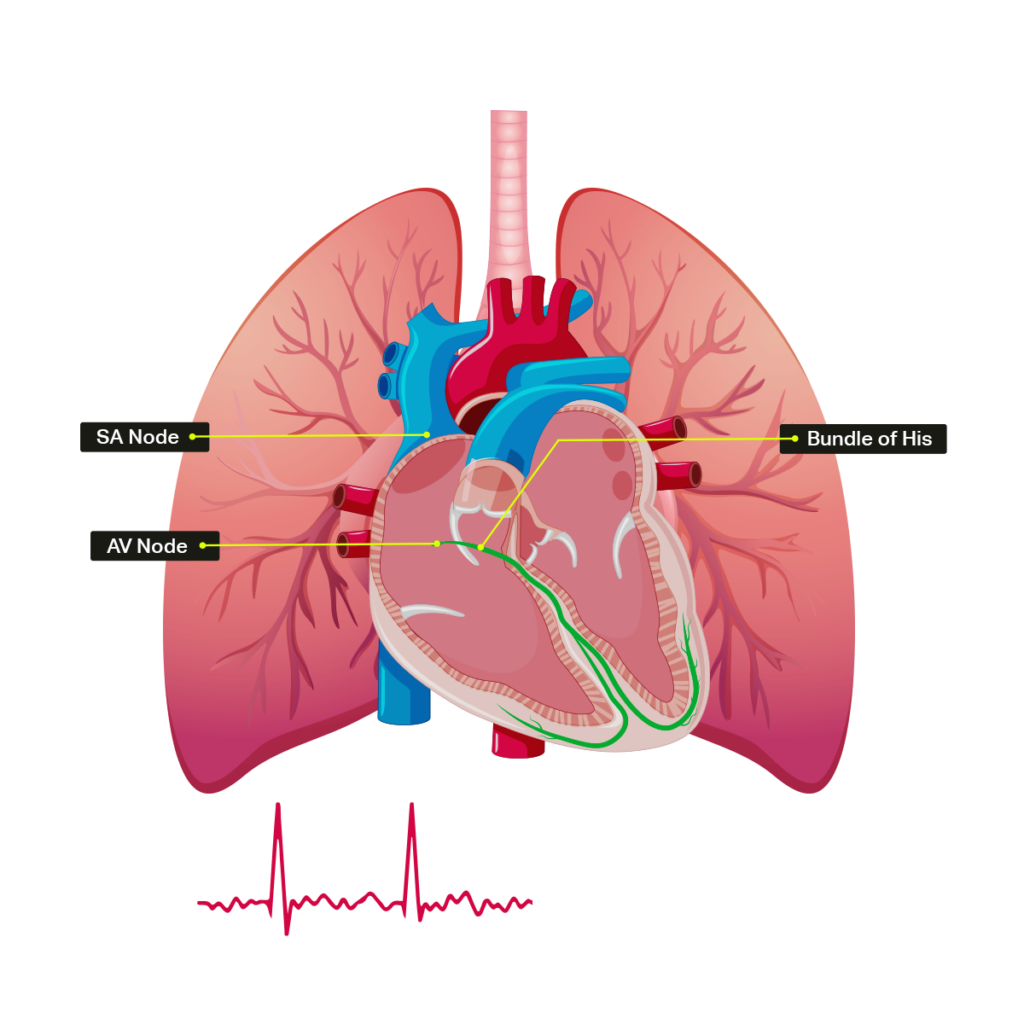
Atrial fibrillation is a problem with the electrical system of the heart.
In atrial fibrillation, there is disorganized, chaotic electric activity in the atria instead of a single electrical signal being sent from the SA node to the AV node with each heartbeat. This causes the atria to beat irregularly and quickly, or to “fibrillate.” This sends several signals to the AV node, which in turn can cause the ventricles to beat quickly and irregularly.
See How the Heart Works for more information on the electrical activity in the heart.
How Common Is Atrial Fibrillation?
Atrial fibrillation is the most common cardiac arrhythmia, affecting about 350,000 Canadians.
Atrial fibrillation becomes more common as we age. Approximately 10% of Canadians over the age of 80 have atrial fibrillation.
Heart failure can also result from atrial fibrillation. Heart failure occurs when your heart is unable to circulate enough blood to meet your body’s needs. Atrial Fibrillation’s irregular, fast heartbeat causes ineffective blood pumping, which, if uncontrolled, can weaken the heart.
What Causes Atrial Fibrillation?
Most common risk factors are older age and high blood pressure for many years. Being overweight, sleep apnea and abnormalities of the thyroid function are also risk factors, as well as genetic factors which are still being studied.
What Is the Risk with Atrial Fibrillation?
Atrial fibrillation causes two main problems:
- Palpitations, fatigue or lightheadedness caused by a very fast heart rate
- Stroke
Fibrillation of the atria can lead to sluggish blood flow, pooling of blood or formation of a blood clot in the atria. If this blood clot is pumped to the brain, it causes a stroke. It is estimated that about 20% of strokes are caused by atrial fibrillation. Unfortunately, the strokes associated with atrial fibrillation tend to be large strokes and often leave patients with significant disability.
How Is Atrial Fibrillation Diagnosed?
Atrial fibrillation is typically diagnosed with an ECG. It may also be detected with a heart monitor that is worn for an extended period of time. These tests may be ordered after an individual has a stroke to determine if atrial fibrillation has caused the stroke.
After atrial fibrillation is diagnosed, other tests are usually considered. An ECHO is usually ordered to make sure that there is no underlying structural problem with the heart (like a weak heart muscle or valve problem). A blood test to check the thyroid function is also usually ordered.
Other tests, like a sleep study, may be considered to see if sleep apnea is contributing to atrial fibrillation.
Are There Different Types of Atrial Fibrillation?
Paroxysmal atrial fibrillation is when a person is in atrial fibrillation for a period of time and naturally returns to normal heart rhythm (called sinus rhythm).
Permanent atrial fibrillation is when someone is in atrial fibrillation all of the time.
How Is Atrial Fibrillation Treated?
Atrial fibrillation can be treated with medications and procedures to reduce its symptoms, control the heart rate, and prevent a stroke.
Treatments include:
- Electrical Cardioversion: Shocking the heart to put it back into the normal rhythm. This is normally done as a day procedure. The patient is put to sleep briefly, and an electrical shock is delivered to the heart.
- Medications to control the heart rate such as beta-blockers, calcium channel blocks, and digoxin. These medications slow the heart rate, but do not put the heart back into the normal rhythm.
- Medications to maintain sinus rhythm: Anti-arrhythmics may be used to try to return the heart to its normal rhythm (sinus rhythm).
- Ablation: An invasive procedure involving the insertion of catheters (thin flexible tubes) in the groin, which are then passed to the heart. Small areas of the heart that are thought to be responsible for atrial fibrillation are then destroyed with these tubes.
- Blood thinners: Blood thinners are used to reduce the risk of stroke in patients with atrial fibrillation.
What Blood Thinners are Available?
An individual’s risk of stroke will help determine if a blood thinner is required and which one is recommended. Most people require a blood thinner.
Aspirin is a mild blood thinner that lowers the risk of stroke in atrial fibrillation by about 20%.
There are more powerful blood thinners that reduce the risk of stroke even further, up to 60-70%.
Warfarin, or Coumadin, is an older medication that is effective at preventing strokes. However, it is not an easy medication to take, as there is not a single correct dose for everyone, and an individual’s dose will usually change over time. Regular blood tests (called an INR) are required to make sure the blood is thin enough, but not too thin. Warfarin also has many interactions with other medications and some foods.
Newer medications called direct oral anticoagulants have been shown to be as effective as warfarin in reducing the risk of stroke and do not require regular blood tests to monitor. Four are currently available in Canada:
- Dabigatran (Pradaxa®)
- Rivaroxaban (Xarelto®)
- Apixaban (Eliquis®)
- Edoxaban (Lixiana®)
These medications are all cleared by the kidneys, so they can’t be used in people with very reduced kidney function. They are also not safe in patients with mechanical heart valves.
What Are the Downsides of Being on A Blood Thinner?
The main downside of being on a blood thinner is the risk of bleeding. If you are on a blood thinner and hit yourself, you will bruise more easily. If you cut yourself, it will take longer to stop bleeding. This type of minor bleeding is not a big concern.
The main concern is major internal bleeding – usually in the gastrointestinal tract (stomach or colon). It will be obvious if you see red blood in your bowel movement that you are bleeding internally. However, if the bleeding is coming from the stomach, it may come out in the bowel movement as a dark black tar. If you notice anything like this, you should immediately seek medical attention.
The other main site of internal bleeding is inside the brain. This is very rare but can be devastating. It may occur after trauma (like a fall) or sometimes spontaneously. Slurred speech or drowsiness may be a sign of this complication and requires immediate medical attention.
There are reversal agents (antidotes) being developed for all the blood thinners mentioned above.
Your healthcare provider will talk to you about which blood thinner option is best for you.
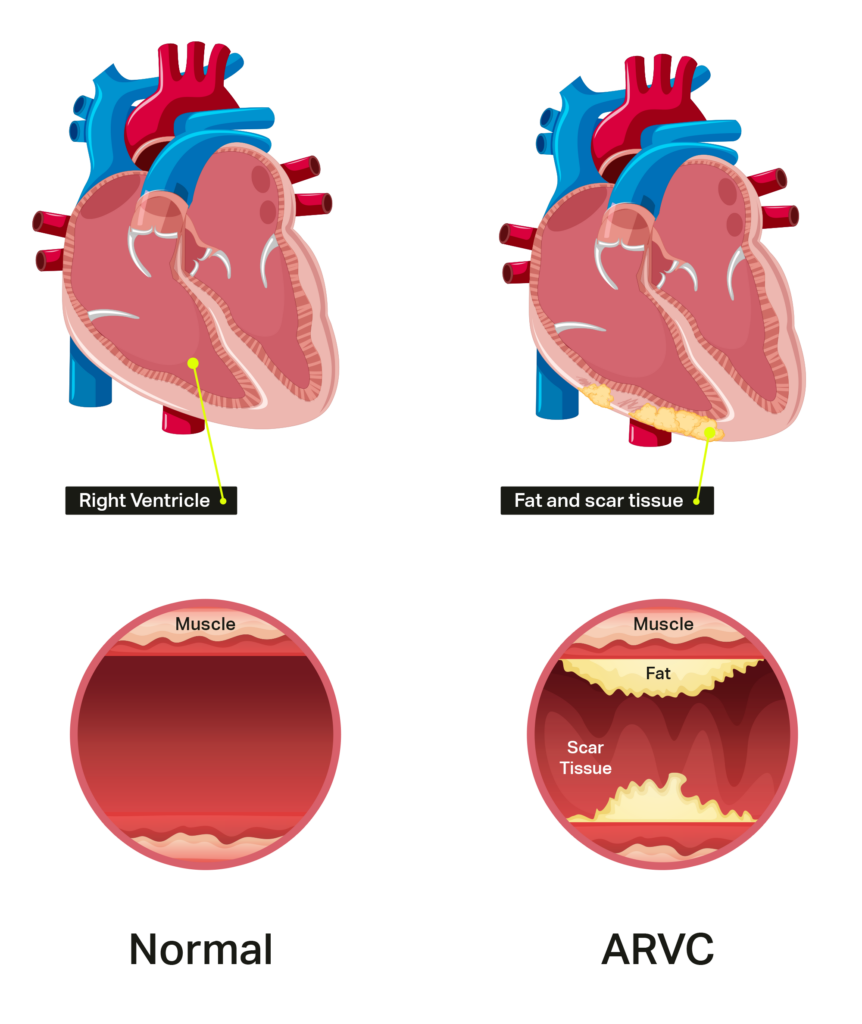
Arrhythmogenic Right Ventricular Cardiomyopathy (ARVC)
It usually affects the right ventricle but may also affect the left ventricle as well.
What Are the Symptoms Of ARVC?
People affected by ARVC may develop symptoms of heart failure because the ventricle becomes weak. This results in fatigue, shortness of breath, and swelling in the legs and abdomen.
Having ARVC also predisposes an individual to arrhythmias. These ventricular arrhythmias may cause someone to experience palpitations, feel dizzy, pass out, or even to die suddenly.
How Is ARVC Diagnosed?
ARVC is diagnosed based on family history and investigations, like an ECG and ECHO. A cardiac MRI is also a good test to look at the right ventricle and see if muscle has been replaced by fat or scar tissue.
Because ARVC is usually an inherited condition, genetic testing via a blood test can sometimes be used to screen family members of affected individuals.
How Is ARVC Treated?
Unfortunately, ARVC cannot be cured. Treatment aims to control the symptoms of heart failure and treat potentially life-threatening arrhythmias with medications or ICD.
If the symptoms of ARVC cannot be controlled with medications and device therapies, patients may be considered for a heart transplant.
People diagnosed with ARVC should not participate in competitive sports.
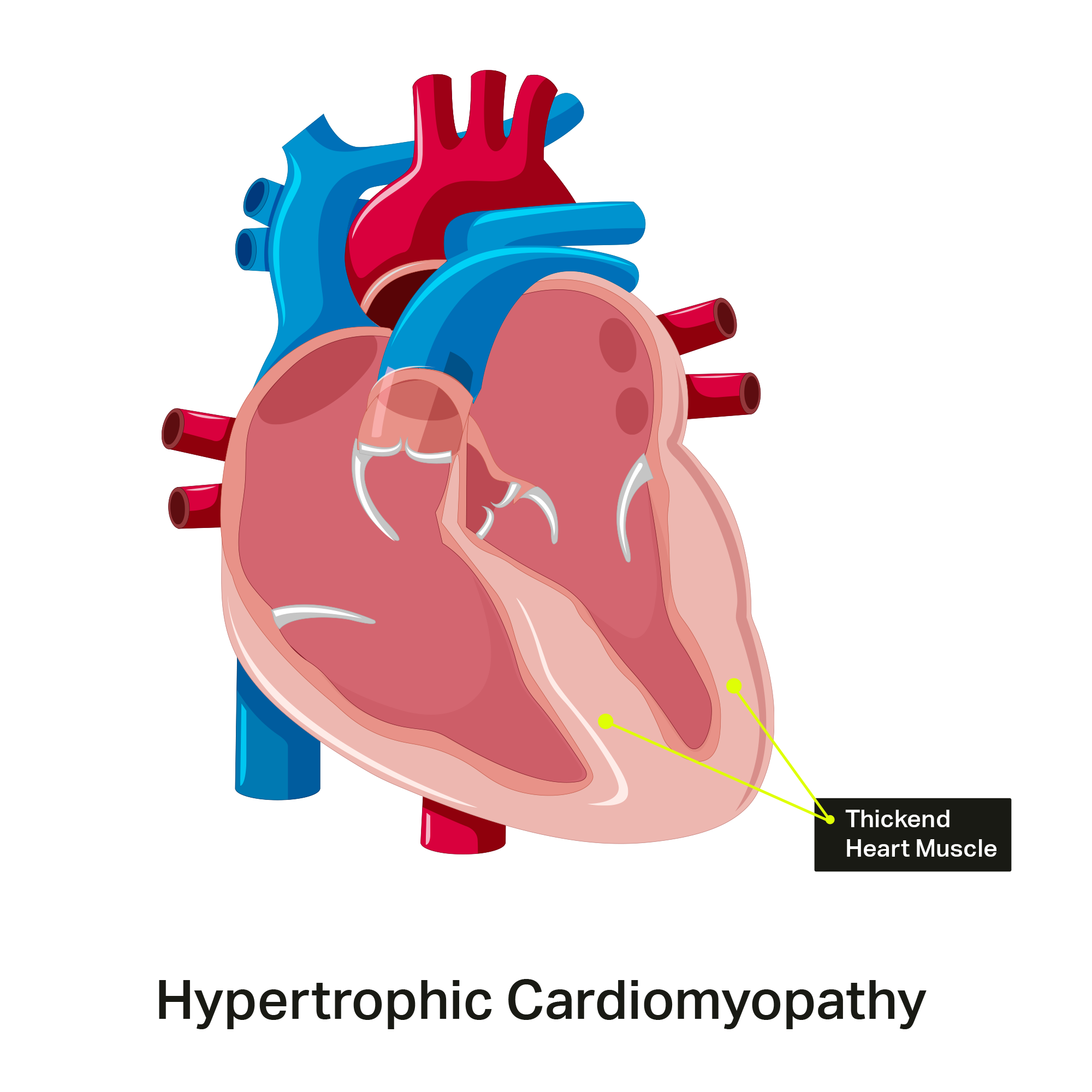
Hypertrophic Cardiomyopathy
Heart muscles can become thickened for other reasons, such as having high blood pressure for many years, but this is not considered HCM.
What are the Symptoms of HCM?
The heart doesn’t relax well if the heart muscle is thickened, so people with HCM can develop problems related to obstruction of blood flow through the heart, arrhythmias, or even heart failure. They may also experience dizziness, chest pain, shortness of breath, palpitations (racing heart) or fainting. When the thickened heart muscle blocks blood flow out of the heart, it is called hypertrophic obstructive cardiomyopathy (HOCM). However, many patients with HCM never develop any symptoms or complications.
HCM is also the most common reason for sudden death in a young person. After being diagnosed, people with HCM should not participate in competitive sports.
If a family member is diagnosed with HCM, other individuals in the family should be screened. Tests to screen for HCM include an ECHO or a blood test to look for a genetic mutation.
How is HCM Treated?
Depending on the severity of the disease, therapies may include medications, invasive procedures (like septal alcohol ablation or surgical myectomy), and devices (like an ICD). Sometimes the disease is severe enough that people may be considered for a heart transplant.
Sarcoidosis
Sarcoidosis is an inflammatory condition that can affect many different organs. It is characterized by collections of inflammatory cells, called granulomas. It is not entirely clear what causes these granulomas to form, but they may be related to the body’s own immune system response to an infection. There is also likely some genetic component, which is why sarcoidosis sometimes runs in families.
Sarcoidosis affects the heart in two main ways as the granulomas can deposit in the heart muscle:
- Problems with the conduction system of the heart, leading to either a slow or fast heart rhythm problem
- A weakened heart muscle, resulting in heart failure
How is Sarcoidosis Diagnosed?
It is difficult to diagnose cardiac sarcoidosis. The diagnosis may be considered in younger people who present with slow heart rhythms. The diagnosis may also be considered if sarcoidosis is found to affect other organs, like the lungs.
The two main tests to diagnose cardiac sarcoidosis are a PET scan and a cardiac MRI. Other tests like an ECG, ECHO, holter monitor, coronary angiogram, or heart biopsy.
How is Sarcoidosis Treated?
Because sarcoidosis is thought to involve the body’s immune system, the treatment is immunosuppressive medications. Corticosteroids (like prednisone) are usually taken, followed by other medications. Other device therapies, like a pacemaker or ICD, may be considered.
Sleep Apnea
Sleep apnea is a condition where people stop breathing during sleep. There are two main forms of sleep apnea:
- Obstructive sleep apnea: The airway collapses during sleep and prevents air from getting to the lungs
- Central sleep apnea: The brain does not send the right signal to the muscles that control breathing
Sometimes people have a combination of both types of sleep apnea.
Common symptoms of sleep apnea are:
- Loud snoring
- Difficulty falling and staying asleep
- Poor quality sleep – waking up and feeling like you have not had a good rest
- Excessive daytime tiredness, feeling the need to nap in the afternoons
- Headaches in the morning
Sleep apnea is very common in people with heart failure. If untreated, it can lead to worsening of the symptoms of heart failure.
How is Sleep Apnea Diagnosed?
Sleep apnea is diagnosed with a test called a sleep study. You will sleep over in a hospital or clinic and be monitored closely during sleep to determine how deeply you are sleeping, if you stop breathing, and how often you stop breathing.
Depending on the type and severity of sleep apnea, different treatments are available such as weight loss or lifestyle changes for mild sleep apnea or a continuous positive airway pressure (CPAP) machine for more severe cases. Your health care provider will talk to you about which is right for you.
Cardiac Amyloidosis
Cardiac amyloidosis is a condition where there is a deposit of abnormal proteins, or amyloid, in the heart tissue. Over time, this causes the heart to become thicker, stiffer, and less effective at pumping blood out to the rest of the body, leading to heart failure.
Cardiac amyloidosis can also affect the electrical pathways in the heart, which can cause abnormal heart rhythms.
Symptoms of cardiac amyloidosis include:
- Shortness of breath with activity or when lying flat in bed
- Low blood pressure
- Fatigue
- Dizziness/Light-headedness
- Feelings of the heart racing or skipping beats
- Swelling to feet/legs/abdomen
A number of tests may be performed to diagnosis cardiac amyloidosis such as ECG, ECHO, cardiac CT scan, coronary angiogram, MRI, PET scan or cardiac biopsy.
In most cases, cardiac amyloidosis is caused by one of two abnormal proteins: light-chain or transthyretin.
Light chain amyloidosis
Plasma cells are located in the bone marrow and produce antibodies for the immune system. Antibodies are made up of heavy and light chains. When the plasma cell becomes malignant it produces too many light chains for that antibody.
This can cause a few different scenarios:
- Monoclonal gammopathy of undetermined significance (MGUS) when abnormal plasma cells are in a small part of bone marrow and removed through the urine.
- Myeloma when abnormal plasma cells take up a bigger area of the bone marrow and can cause high calcium levels (hypercalcemia), low hemoglobin (anemia), kidney dysfunction, and bone damage.
- Light chain (AL) amyloidosis: Abnormal plasma cells produce light chains that fold abnormally and bind together forming amyloid particles. These amyloid particles are found in the blood and can be deposited into the heart or other organs.
AL amyloidosis is the most commonly diagnosed form of systemic amyloidosis, generally affecting people between the ages of 40-85 years.
Transthyretin amyloidosis
Transthyretin is a protein produced by the liver. Some transthyretins can fold abnormally and become amyloid deposits. There are two types of transthyretin (ATTR) amyloidosis: wild-type ATTR and mutant ATTR.
Wild-type can create amyloid deposits over several years and is more common in people over the age of 70 years. Amyloid deposits form primarily in the heart, veins and arteries, and soft tissues.
In mutant type ATTR, people are born with an abnormal transthyretin gene leading to more rapid amyloid deposition. The heart and nerves are the most common sites for amyloid deposits in mutant ATTR.

 Sharon Bray
Sharon Bray
 Paula Henderson
Paula Henderson
 Isabel Victal
Isabel Victal
 Cindy
Cindy